
BioFlo PICC with Endexo Technology
BioFlo PICC
Making a Material Difference
The BioFlo PICC is the first PICC of its kind with Endexo Technology, providing a catheter more resistant to the accumulation of blood components (platelets and thrombus).
· The BioFlo PICC is the only PICC manufactured with Endexo Technology, a permanent and non-eluting integral polymer.
· In-vitro blood loop model test results show that on average, the BioFlo PICC with Endexo Technology has 87% less thrombus accumulation on its surface compared to commonly used PICCs based on platelet count.†
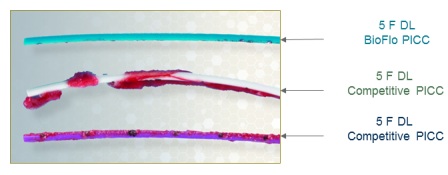
· Endexo Technology has been shown to be effective in reducing thrombus accumulation. The reduction in thrombus accumulation was evaluated using in-vitro and in-vivo models. Pre-clinical in-vitro and in-vivo evaluations do not necessarily predict clinical performance with respect to thrombus formation.
· The BioFlo PICC is available with PASV Valve Technology, making it the first catheter that combines the reduced thrombus accumulation benefit with our patented valve designed to automatically resist backflow and reduce blood reflux on the inside of the catheter. The BioFlo PICC is also available in non-valved configurations.
† Based on benchtop test results which may not be indicative of clinical results. Data on file.
Endexo Technology
Make Protection Part of the Mix
Endexo Technology is a proprietary polymer that is present throughout the catheter shaft material and remains present for the life of the catheter, providing long-term durability.††
BioFlo Technology is not a coating or impregnated into the catheter. Instead, the Endexo technology polymer is added directly to the base polyurethane during the manufacturing process. Endexo technology is present throughout the catheter material, including the outer surface, inner surface and even the cut catheter tip.
BioFlo Technology does not contain agents that may be associated with bacterial resistance, including antibiotics or antimicrobials. In addition, this catheter does not contain heparin, which may help minimize the complications associated with heparin use.
†† Technical data on file.
PASV Valve Technology
The Power of PASV
PASV stands for Pressure Activated Safety Valve which is proprietary to AngioDynamics. The PASV Valve is a direction-specific valve located in the proximal hub of the catheter, outside of the patient and the bloodstream. It is designed to reduce blood reflux into the catheter lumen that could lead to catheter-related complications and has over 10 years of clinical experience.

The PASV Valve product does not have clamps or require a specific needleless connector. The PASV Valve may be maintained with a minimum weekly saline flush.†††
†††Clinicians are instructed to follow institutional protocols concerning PICC maintenance. Heparin may be used based on the physician’s clinical assessment of patient needs or per institutional protocols.
BioFlo PICC
with Endexo and PASV Valve Technology
Providing Total Catheter Performance
Large Lumen Diameters
· Power injectable catheter designed to provide greater flow rates at lower pressures
· Developed to minimize risk of catheter occlusion
· Designed for easy blood withdrawal
Simplified Care and Maintenance
· Minimum weekly saline flush recommended on valved catheters
· Clampless extension legs on valved catheters
· Freedom to choose your preferred needleless connector
Ease of Placement
· Variety of kit configurations
· Exact-Length Measurement System
· Trimmable catheter tip
· Radiopaque
· Short reverse taper extrusion
Flow Rates
· Maximum flow rates range from 1 mL/sec to 6 mL/sec, based on catheter size and configuration. Refer to product labeling for more detailed information.
Value Added Programs and Services
Convenience Kit Program
Improve clinician efficiency, productivity and cost savings with our Convenience Kit Program. Our comprehensive Program provides clinicians with solutions to streamline PICC placement procedures, eliminate the cost of unnecessary supplies and meet department budget guidelines. In addition, clinicians are able to choose from our broad portfolio of PICCs for a variety of placement settings, insertions techniques and clinical applications.
Clinical Education
AngioDynamics retains a highly credentialed team of clinical specialists committed to providing educational support and training. In addition, a wide range of continuing education programs and support materials are available to you, including wall charts and patient education materials, all designed to reinforce best practices for catheter insertion, care and maintenance.
Technical Support for this product and other AngioDynamics’ Vascular Access (Show Info in box) products is available 24 hours a day by calling our Vascular Access Product Reference Line at 800.513.6876
Important Risk Information
INTENDED USE/INDICATIONS FOR USE: The BioFlo PICC with Endexo and PASV Valve Technology is indicated for short or long-term peripheral access to the central venous system for intravenous therapy, including but not limited to, the administration of fluids, medications and nutrients; the sampling of blood; and for power injection of contrast media. CONTRAINDICATIONS: Venous thrombosis in any portion of the vein to be catheterized. Conditions that impede venous return from the extremity such as paralysis or lymphedema after mastectomy. Orthopedic or neurological conditions affecting the extremity. Anticipation or presence of dialysis grafts or other intraluminal devices. Hypercoagulopathy unless considerations are made to place the patient on anticoagulation therapy. Pre-existing skin surface or subsurface infection at or near the proposed catheter insertion site. Anatomical distortion of the veins from surgery, injury or trauma. Inadequate antecubital veins. Anatomical irregularities (structural or vascular) which may compromise catheter insertion or catheter care procedures. WARNINGS: Due to the risk of exposure to bloodborne pathogens, care providers must adhere to guidelines for universal blood and bodily fluid precautions in the care of all patients. Sterile technique must be strictly adhered to during any handling of the device. Contents are supplied sterile by EO for single patient use only. Do not use if sterile barrier is damaged. Do not use if product has been damaged. Do not reuse, reprocess or resterilize, to do so may compromise device integrity and/or lead to device failure which in turn may result in patient injury, illness or death; and may also create a risk of contamination, patient infection or cross infection which may lead to injury, illness or death of the patient. Do not place the catheter into the right atrium or the right ventricle of the heart. Do not attempt to trim the catheter with the guidewire or stylet loaded as catheter, stylet, or guidewire may become damaged resulting in patient injury. Failure to warm contrast media to body temperature prior to power injection may result in catheter failure. Failure to ensure patency of the catheter prior to power injection studies may result in catheter failure. Power injector’s pressure limiting (safety cut-off) feature may not prevent over-pressurization of occluded catheter. Exceeding the maximum allowable flow rate (per the Directions for Use) may result in catheter failure and/or catheter tip displacement. Catheter indication for power injection of contrast media implies the catheter’s ability to withstand this procedure, but does not imply appropriateness of this procedure for a particular patient. A trained clinician is responsible for evaluating the health status of a patient as it pertains to a power injection procedure. The maximum pressure of power injectors used with the BioFlo PICC must not exceed 325 psi. For triple lumen catheters, only the purple lumen is for power injection. Do not use lumen marked “No CT” for power injection of contrast media as it may result in catheter damage or patient injury. PRECAUTIONS: Do not insert the stiff end of the floppy-tipped guidewire into the vein. Acetone and polyethylene glycol-containing ointments should not be used with polyurethane catheters, as these may cause failure of the device. Following institutional policy, secure catheter externally to prevent catheter movement, migration, damage, kinking or occlusion. It is recommended that institutional protocols be considered for all aspects of catheter use consistent with the instructions provided herein including flushing of occluded catheters and power injection. The BioFlo PICC catheter testing included 10 power injection cycles. Use of a needle to access the catheter is not recommended. However, if a needle is used, do not use a needle longer than 1.9 cm as it may cause damage to the valve. Do not reinsert stylet into catheter, as damage to valve, catheter and vein may result. If a needleless connector is attached to catheter hub, first ensure that it will sustain power injection.When inserting a triple lumen catheter, the power injectable lumen must be used for guidewire/stylet placement. Refer to Directions for Use provided with the product for complete instructions, warnings and precautions. CAUTION: Federal Law (USA) restricts this device to sale by or on the order of a physician.
